Glaucoma – Canine
Rachel L. Davis1
VIN Publication
Abstract
Glaucoma refers to elevated intraocular pressure (IOP) that is either sustained or fluctuating. It can ultimately result in death of retinal ganglion cells. Glaucoma encompasses a broad category of diseases but is commonly referred to in the singular tense. Although normotensive glaucoma has been identified in people, it has not been documented in the cat or dog.
Learn about glaucoma in dogs, including causes, symptoms, diagnosis, and treatment. Understand the difference between primary and secondary canine glaucoma and available management options below.
Keywords: Glaucoma in dogs, glaucoma, ocular, pressure, intraocular, tonometry, blindness, uveitis, cornea, retina, optic, nerve, aqueous, humor, surgery, prosthesis, dog, cat, canine, feline, veterinary, ophthalmology, gonioscopy, buphthalmos, IOP, lens, luxation, cataract, iridocorneal, angle, pigmentary, melanocytic, prostaglandin, analogues, mannitol, dorzolamide, latanoprost, brinzolamide, enucleation, goniodysgenesis, breeds, therapy, diagnosis, treatment, management
Contributors:
1Revised by Rachel Davis DVM, MS, DACVO at Animal Eye Clinic, Westfield, Indiana, USA, on 04/05/2022
Revised by Thomas Chen DVM, MS, DACVO and Rhea V. Morgan DVM, DACVO, 9/14/20161, 2
Original author was Ian P. Herring DVM, MS, DACVO, 9/9/2004
Correspondence:
Rachel L. Davis, DVM, MS, Diplomate, ACVO – Ophthalmologist
Animal Eye Clinic
4750 Killarney Drive
Carmel, IN 46033
Email: info@indyaec.com
Pathophysiology
Glaucoma is one of the most damaging of all ocular diseases. Glaucoma initially induces microscopic damage that can lead to macroscopic changes as the disease progresses. Glaucoma is ultimately a neurodegenerative disease. Because retinal neuronal cells affected by elevated IOP are responsible for relaying visual signals, glaucoma often results in blindness. Glaucoma also has deleterious effects on other ocular structures including sclera, cornea, iridocorneal angle, iris, lens, vitreous, and optic nerve.
Classifications
Primary Glaucoma
Primary glaucoma is inherited, breed-related glaucoma. It typically affects purebred dogs, and is rare in cats. Primary glaucoma develops from anatomic malformation of the iridocorneal drainage angle (e.g. narrow angle glaucoma, goniodysgenesis), or from accumulation of abnormal glycosaminoglycans in the trabecular meshwork that results in resistance to aqueous humor outflow (i.e. primary open angle glaucoma, POAG). Breeds with narrow or closed-angle glaucoma also typically have developmental goniodysgenesis. Anatomic conditions responsible for primary glaucoma are bilateral, although onset of glaucoma is almost always unilateral and asynchronous. If one eye is diagnosed with primary glaucoma, the contralateral eye is at risk for developing the disease.
Secondary Glaucoma
Secondary glaucoma is caused by an underlying ocular or systemic disease that results in abnormal aqueous humor drainage from the eye. Potential causes include anterior lens luxation, uveitis, hyphema, pupillary block (e.g. posterior synechiae with iris bombe, lens luxation, vitreous herniation), intraocular neoplasia, pigmentary uveitis, and intraocular surgery. Aqueous outflow obstruction may also occur with pre-iridal fibrovascular membrane formation, peripheral anterior synechia, and cellular infiltration of the iridocorneal angle. Secondary glaucoma is often unilateral, although bilateral glaucoma may occur if the underlying disease afflicts both eyes.
Disease Description in This Species:
Etiology
Genetic studies have pinpointed various genes responsible for development or increased susceptibility to glaucoma in some dog breeds. Examples include primary closed angle glaucoma (PACG) in the shiba inu and shih tzu; POAG in the beagle (autosomal recessive trait) and Norwegian elkhound; melanocytic glaucoma in the Cairn terrier (autosomal dominant); and primary lens luxation in several terrier breeds.1-9 In the beagle, a single base pair substitution (chr20:53096339 (canFam3): G>A) on ADAMTS10 gene is the responsible mutation.4 In the Norwegian elkhound, a different base pair substitution (c.1441G>A p.Ala387Thr) on chromosome 20 of ADAMTS10 is responsible.5
Diagnosis
Physical Examination Findings/History: Because glaucoma may arise secondary to uveitis associated with infectious and systemic diseases, as well as metastatic intraocular neoplasia, systemic abnormalities are possible. Complete physical examination is indicated to identify the presence of systemic disease and its contribution to development of glaucoma. In many cases, however, glaucoma is primarily an ocular condition and identified on ophthalmic examination.
Ophthalmic Examination Findings: Changes associated with glaucoma vary depending on the stage of disease. Findings associated with acute glaucoma may include blepharospasm, conjunctival hyperemia, episcleral injection, ciliary flush (perilimbal corneal vascularization), diffuse corneal edema, mydriasis, vision loss, and optic nerve and peripapillary retinal edema. With chronicity, additional findings may include buphthalmos, corneal Haab’s striae, lens subluxation, cataract, optic nerve cupping and atrophy, and retinal degeneration. Some eyes have more subtle abnormalities (i.e. “silent glaucoma”). Thorough ophthalmic examination is indicated to search for potential causes of glaucoma and assess the opposite eye. Referral for gonioscopy, slit lamp biomicroscopy, and indirect ophthalmoscopy should be considered.
Tonometry: Diagnosis of glaucoma relies on measurement of IOP via tonometry. IOP can be reliably measured by indentation (e.g. Schiotz tonometer), applanation (e.g. TonoPen), and rebound tonometry (e.g. TonoVet). Schiotz tonometry is technically challenging and has been largely replaced by rebound or applanation tonometry because the latter are now readily available. Digital estimation of IOP (i.e. pressing on the globe with one or more fingers) is unreliable. Topical ophthalmic anesthetic is applied prior to indentation or applanation tonometry but is not needed for rebound tonometry. Because normal IOP can vary between individuals, IOP differences between the two eyes are as important as absolute IOP values. Always perform tonometry on both eyes. Normal IOP in the dog generally ranges from 10-20 mmHg but can be as high as 20-24 mmHg with rebound tonometry. IOPs >25 mmHg or a significant disparity in IOP (i.e. >7-8 mmHg) between eyes is considered abnormal. It is important to note that a single measurement of IOP may not accurately reflect aqueous humor dynamics because IOP may vary widely during the day, and from one day to the next. Animals with glaucoma have sustained elevation of IOP.
IOP can be falsely elevated with patient excitement, fear, or stress; excessive physical restraint (especially pressure applied to the head and neck); marked blepharospasm with globe retraction; and application of digital pressure to the eye while holding the eyelids open. If IOP results are unexpected or do not match the clinical signs, repeated measurements can be performed in a darkened, quiet environment using different restraint techniques. Note that almost all sedatives alter IOP, although these alterations are not typically clinically relevant.
Gonioscopy: Gonioscopy is visualization of the iridocorneal angle through a specialized corneal contact lens (Figure 1). The iridocorneal angle of the dog is not readily visible because of its steep corneal curvature; angle of light refraction by the cornea; prominent limbus; and shallowness of the anterior chamber. Iridocorneal angle of the cat can be visualized with indirect ophthalmoscopy and magnification because anterior chamber deeper. Determining morphology of the iridocorneal angle is useful in distinguishing primary from secondary glaucoma.
Goniodysgenesis: Most glaucoma in dogs arises from goniodysgenesis. Goniodysgenesis occurs from failure of rarefaction of tissue in the iridocorneal angle during normal neonatal development, not from development of abnormal tissue later in life. During the first 8 weeks of life, tissue in the normal iridocorneal angle undergoes morphologic changes that cause it to break apart or disintegrate, thereby forming pectinate ligaments separated by open spaces (Figure 2). If this process does not occur, goniodysgenesis results. On gonioscopy, instead of individual pectinate ligaments separated by obvious flow-through openings, large sheets of tissue are separated by irregularly-spaced holes (Figure 3). Even though the iridocorneal angle is structurally abnormal early in life, primary glaucoma typically does not develop until the dog is middle-aged. The reason for this late onset is unclear. Iridocorneal morphologic changes may worsen over time but if pectinate ligaments are normal in a young dog, goniodysgenesis does not develop later.
Angle Closure: Gradual narrowing of the iridocorneal angle occurs that results in closure of the angle within the first 2-3 years of life.11 This narrowing is visible on gonioscopy because the base of the iris lies adjacent to the posterior limbus of the cornea (Figure 4). Collapse of the ciliary cleft and trabecular meshwork also occurs.11 Goniodysgenesis is invariably present in dogs with closed angles but cannot be visualized because the base of the iris obscures the view of the pectinate ligaments.12-16 In clinical practice, PACG associated with angle closure and goniodysgenesis occurs much more often than POAG.
Open Angle: POAG is characterized by a morphologically-normal iridocorneal angle (Figure 2), and development of glaucoma earlier in life than with PACG. Despite its normal appearance, immunohistochemical and microscopic abnormalities of the angle are present. POAG is the least common form of primary glaucoma in clinical practice but has been demonstrated in several dog breeds.
Genetic Testing: DNA tests for POAG are available for the beagle in the USA through Paw Print Genetics, UCDavis Veterinary Genetics Laboratory, and Animal Genetics. In Europe, beagles can be tested for POAG through Genomia and various breeds can be tested through LABOGEN. DNA testing is available in the UK for the basset hound, petit basset fauve de Bretagne, beagle, Norwegian elkhound, and petit basset griffon vendeen through LABOKLIN, and for the beagle through Animal Genetics UK. In Australia and New Zealand, Orivet can test for POAG in the beagle, Norwegian elkhound, and basset hound.
Other Tests: If glaucoma is secondary and may be caused by a systemic illness that has induced intraocular disease, then a thorough medical work is indicated. Although biochemical markers have been found in some human forms of glaucoma, no clinically-applicable markers have been identified in affected dogs and cats.10
Prevalence and Signalment
Overall prevalence of primary glaucoma in purebred dogs is estimated to be 0.89%, and it may approach 5% in some breeds.17 Breeds predisposed to PACG and goniodysgenesis include the akita, American cocker spaniel, basset hound, chow chow, bouvier des Flandres, English cocker spaniel, English springer spaniel, flat-coated retriever, Great Dane, Leonberger, toy and miniature poodle, samoyed, Chinese shar pei, shiba inu, Siberian husky, shih tzu, and Welsh springer spaniel.1-3,12-19 Breeds predisposed to POAG include the beagle, Norwegian elkhound, and petit basset griffon vendéen.20 Breeds predisposed to melanocytic/pigmentary glaucoma include the Cairn and Scottish terrier. Other breeds predisposed to glaucoma but with poorly or undefined types include the Alaskan malamute, Boston terrier, bull mastiff, dalmatian, giant schnauzer, greyhound, Italian greyhound, miniature pinscher, smooth-haired fox terrier, and wirehaired fox terrier.
In many studies published after 1973, females are overrepresented at a ratio of 2:1 to males. In Siberian huskies, eyes with blue irises are more likely to be affected. Age of onset varies depending upon the type and underlying cause. Most dogs with breed-related glaucoma are middle-aged at the time of onset.
Clinical Signs
Signs of acute glaucoma may include blepharospasm, episcleral injection, ocular discharge, diffuse corneal edema, and mydriasis (Figure 5). With unilateral disease, vision loss may not be detected by the owner. Because IOP often fluctuates widely in affected dogs prior to onset of sustained glaucoma, consider referring dogs in at-risk breeds with a history of fluctuating redness or third eyelid elevation (Figure 6) for gonioscopy. These episodes can be misinterpreted as conjunctivitis. Because glaucoma is thought to cause headache-like pain, some dogs may show subtle behavioral signs, such as lethargy, somnolence, and decreased activity. Perilimbal corneal vascularization, aqueous flare, iris discoloration, and dyscoria may be present with underlying intraocular inflammation (Figure 7).
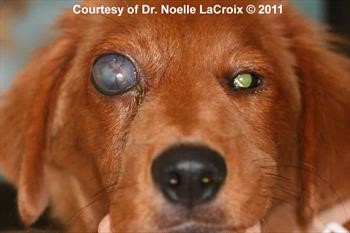
Signs of chronic glaucoma may also include buphthalmos (Figure 8), corneal Haab’s striae (Figure 9), lens subluxation (Figure 10), cataract, retinal and optic nerve atrophy (Figure 11). In general, presence of buphthalmos (i.e. corneal diameter between 9 and 3 o’clock larger than the opposite eye) is an indication glaucoma has been present for some time and the eye is permanently blind. Interestingly, IOP may improve in buphthalmic eyes during end-stage glaucoma as the ciliary body dies and the globe becomes stretched (Figure 12); however, during this process, the patient is painful due to prolonged elevated intraocular pressure. Other ophthalmic and systemic signs may be present if glaucoma is secondary.
Etiology:
- Cataract, intumescent
- Congenital anomalies
- Genetic, hereditary
- Hyphema
- Intraocular neoplasia
- Lens instability
- Lens luxation (anterior)
- Ocular melanosis
- Pupillary block
- Surgery
- Trauma
- Uveitis
Breed / Species Predilection:
- Akita
- Alaskan malamute
- American cocker spaniel
- Basset hound
- Beagle
- Boston terrier
- Bouvier des Flandres
- Cairn terrier
- Chinese shar pei
- Chow chow
- Dalmatian
- English cocker spaniel
- English springer spaniel
- Flat-coated retriever
- Giant schnauzer
- Golden retriever
- Great Dane
- Greyhound
- Italian greyhound
- Leonberger
- Mastiff, bull
- Miniature pinscher
- Miniature poodle
- Miniature schnauzer
- Northern Inuit dog
- Norwegian elkhound
- Petit basset griffon vendeen
- Poodle
- Samoyed
- Scottish terrier
- Shiba inu
- Shih tzu
- Siberian husky
- Smooth fox terrier
- Toy poodle
- Welsh springer spaniel
- Welsh terrier
- Wirehaired fox terrier
Sex Predilection:
Female
Age Predilection:
Mature, middle-aged
Clinical Findings:
- AFEBRILE
- Anisocoria, pupils unequal
- Aphakic crescent
- Behavior abnormal, behavioral change
- Blepharospasm, eye pain
- Blindness acute, sudden
- BLINDNESS OR OTHER VISUAL DEFICIT
- Blindness partial, visual deficit
- Buphthalmos
- Conjunctival congestion, hyperemia
- CONJUNCTIVITIS
- Corneal mass
- Corneal opacity
- Corneal ulcer, keratitis
- Corneal vascularization
- Depression, lethargy
- Dyscoria
- Edema conjunctiva, chemosis
- Edema corneal
- EDEMA, SWELLING
- Epiphora, lacrimation increased
- Episcleral injection/congestion
- Glaucoma
- HYPERPIGMENTATION
- Hypopyon
- Iridodonesis
- Iris bombe
- Iris color variation
- Iris pigmentation abnormal
- Lagophthalmos
- Lens luxation, subluxation
- MASS
- Menace response absent or decreased
- Miosis
- MYDRIASIS
- OCULAR DISCHARGE
- Ocular discharge mucoid
- Ocular discharge serous
- PAIN
- PHOTOPHOBIA
- Pupillary light reflex absent
- Pupillary light reflex decreased
- Sleep inappropriate or abnormal
- Synechia, anterior
- Third eyelid inflammation
- Third eyelid, nictitating membrane prolapsed
- UVEITIS
- Uveitis, anterior
| Diagnostic Procedures: | Diagnostic Results: | |
| Gonioscopy | Mesodermal goniodysgenesis | |
| Narrow, closed iridocorneal angle in eye | ||
| Open iridocorneal angle in eye | ||
| Ocular examination | Anterior chamber deep | |
| Anterior chamber shallow | ||
| Aqueous flare | ||
| Cataract, lens opacity | ||
| Hyphema, blood anterior chamber eye | ||
| Intraocular pressure increased on tonometry | ||
| Iridocyclitis, iris/ciliary body inflamed | ||
| Lens in posterior chamber | ||
| Leukocoria, leukokoria | ||
| Optic disc atrophied | ||
| Optic disc cupping | ||
| Peripapillary tapetal hyperreflectivity | ||
| Retinal blood vessels tortuous | ||
| Retinal degeneration | ||
| Retinal vascularity decreased | ||
| Tapetal reflectivity increased |
Images:

Figure 1. Koeppe goniolens placed on the cornea 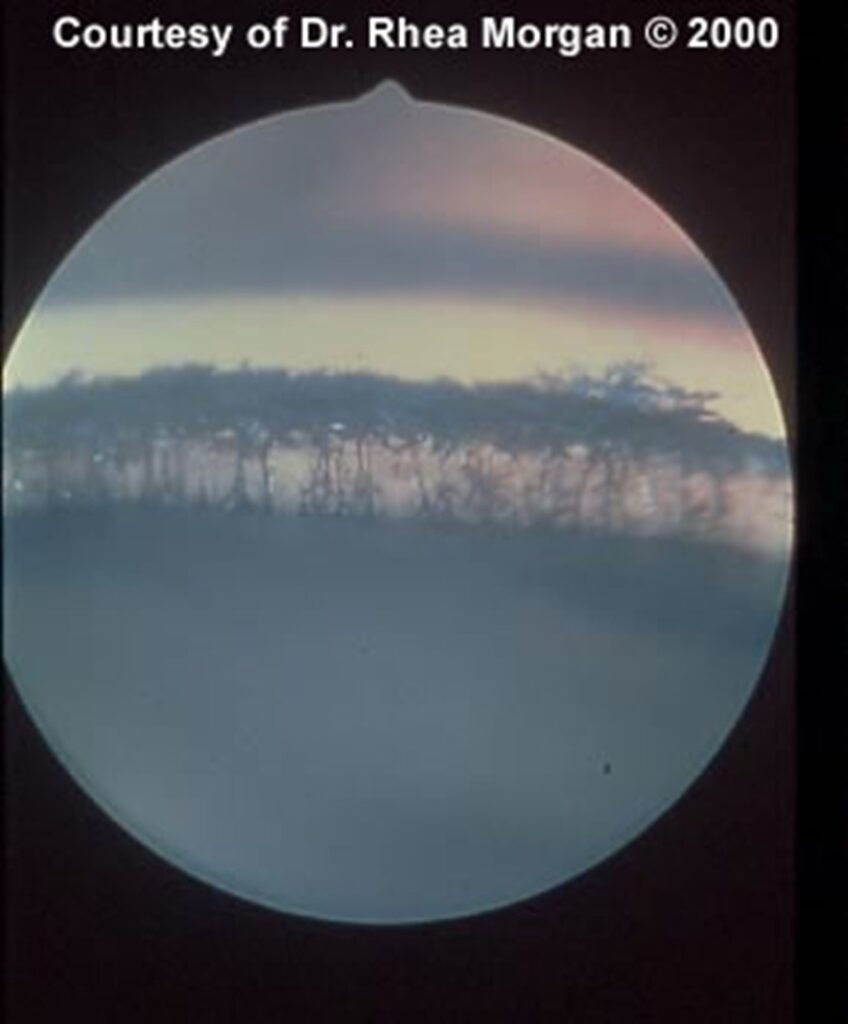
Figure 2. Normal, open angle on gonioscopy.
Thin strands for pectinate ligaments cross the iridocorneal angle between the base of the iris ventrally and the cornea dorsally. Large spaces are present between them.
Figure 3. Goniodysgenesis on gonioscopy.
Instead of pectinate ligaments, the iridocorneal angle is covered with large sheets of tissue broken up by irregularly-shaped holes. Pupil is visible ventrally near flash artifact. Cornea is dorsal to the angle.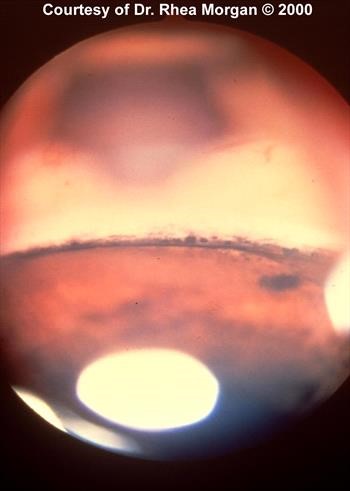
Figure 4. Narrow or closed angle on gonioscopy.
The base of the iris lies close to the cornea and the iridocorneal angle is not visible.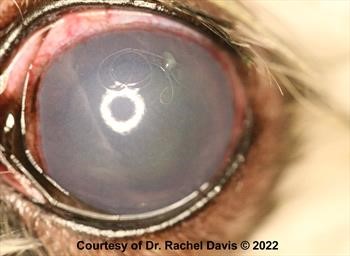
Figure 5. Acute glaucoma in a dog.
Note diffuse corneal edema, scleral injection, conjunctival hyperemia and mydriasis.
Figure 6. Clinical glaucoma in a dog.
Note diffuse corneal edema and third eyelid elevation.
Figure 7. Acute glaucoma in an eye with a blue iris.
Note iris color change, as well as, perilimbal corneal vascularization, diffuse corneal edema, and conjunctival hyperemia.
The horizontal lines are Haab’s striae and are due to breaks in Descemet’s membrane. They are secondary to elevated eye pressure. Primary glaucoma was suspected in this dog. Click here to see board discussion. 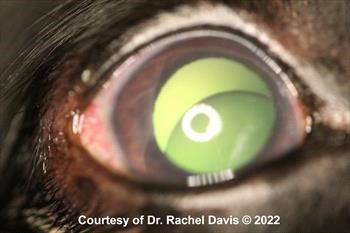
Figure 10. Ventronasal lens subluxation in a glaucomatous eye.
Enlargement of the eye caused zonules to break around the circumference of the lens, allowing it to subluxate.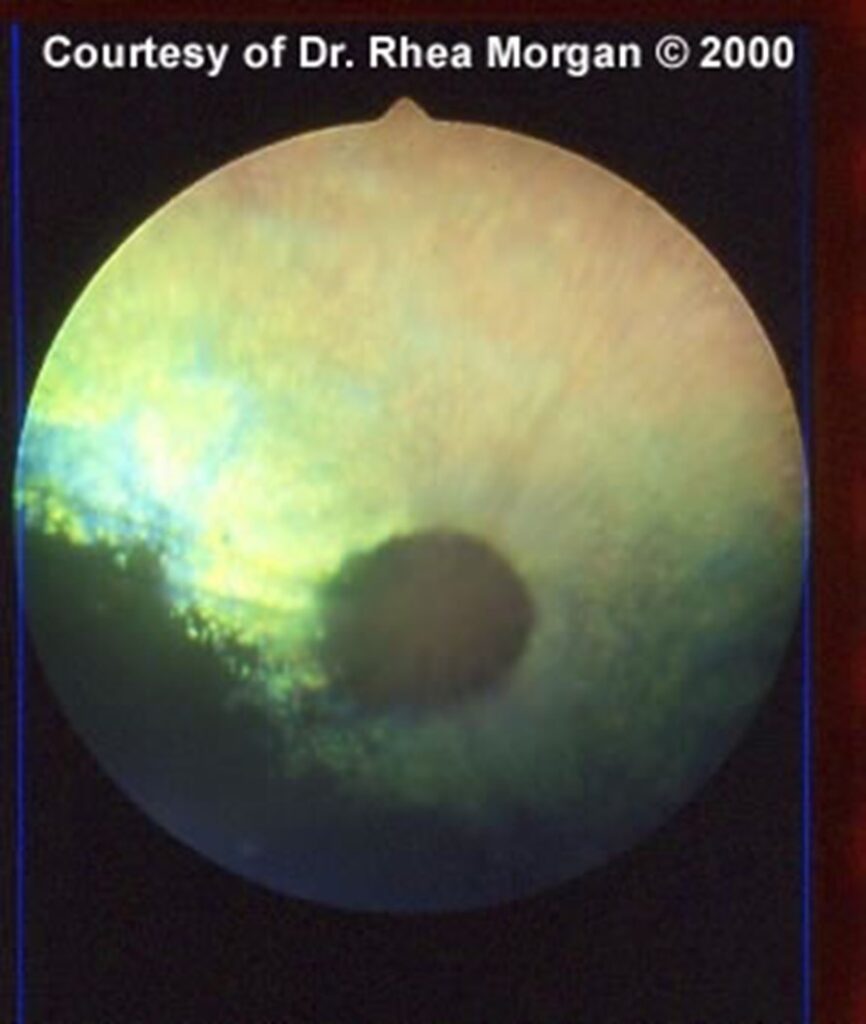
Figure 11. Complete retinal and optic nerve atrophy secondary to glaucoma. 
Figure 12. Normotensive buphthalmic eye.
Click here to see board discussion.
Figure 13. Iridencleisis 14 days postoperatively.
Eye developed glaucoma following phacoemulsification of a cataract, and remains visual and comfortable.
Figure 14. Iridencleisis 10 days postoperatively.
Eye was diagnosed with PACG and treated prophylactically with medication for 3.5 years. Iridencleisis was performed when IOP became elevated. Eye remains visual, with normal IOP.
Treatment / Management:
Glaucoma is often an aggressive, progressive disease. Clinical suspicion of primary glaucoma warrants immediate referral to a veterinary ophthalmologist for treatment of the affected eye and evaluation of the opposite eye. Unfortunately, pain associated with onset of glaucoma may be overlooked because blepharospasm and photophobia do not always occur. Hence, glaucoma can be advanced before it is diagnosed. Treatment of glaucoma can be subdivided into therapy of acute, severe elevation of IOP; medical management of chronic glaucoma; surgical therapy for sighted eyes or eyes with the potential for sight; and surgical therapy of chronic, unremitting glaucoma in blind eyes. Potentially visual eyes with primary glaucoma, and eyes with certain forms of secondary glaucoma (e.g. anterior lens luxation, neoplasia) are usually treated with surgery or other interventions because medical therapy alone is often unsuccessful.
SPECIFIC THERAPY
Acute Glaucoma Therapy
It is important that IOPs >50 mmHg be lowered rapidly (i.e. within 6-12 hrs) in an attempt to save vision. In cases of primary glaucoma, one of the prostaglandin analogues (e.g. latanprost, bimatoprost, travoprost, tafluprost) may be given q 10-15 mins for 2-3 doses. It is important to rule out secondary glaucoma because prostaglandin analogues upregulate inflammation and cause potent miosis. Patients with uveitis or anterior lens luxation should not receive prostaglandin analogues topically.
If IOP does not fall within 40-45 minutes, then systemic hyperosmotic medications may be administered. Such medications include mannitol (1-2 g/kg IV slowly over 20-40 min) or glycerin (1-2 Gm/kg PO). With administration of hyperosmotic agents, water intake is initially restricted and then slowly reintroduced. A second dose of mannitol can be given an hour later, if IOP does not decrease with the initial dose. Administration of >2 doses of mannitol may cause rebound ocular hypertension when water is reintroduced. Hyperosmotic agents can be administered to transiently lower IOP while patients are being referred for more interventive therapy, such as glaucoma surgery and removal of an anteriorly luxated lens. Hyperosmotic agents have very transient effects (<8-12 hrs), so are not used for chronic management of glaucoma. Hyperosmotic agents are contraindicated in animals with congestive heart failure, renal insufficiency, and dehydration. In addition, glycerin may cause vomiting and is converted to glucose, so it should be avoided in dogs with diabetes mellitus.
Maintenance Medical Therapy
Various anti-glaucoma medications are available, most of which are topical formulations. Choice of medication is somewhat dependent on the type of glaucoma; whether the eye is visual or has a good prognosis for vision with IOP control; client goals; and concurrent systemic or ocular conditions. Medical therapy may be the primary method of treating many forms of secondary glaucoma. For eyes with primary glaucoma, combination of medical and surgical therapy results in the best outcomes. Glaucoma secondary to intraocular surgery (e.g. phacoemulsification, lensectomy) may also require a combination of medical and surgical therapy depending on response to initial medications and other factors. Several different classes of anti-glaucoma medications are available.
Carbonic Anhydrase Inhibitors (CAIs): Topical CAIs include dorzolamide 2% and brinzolamide 1%. Dorzolamide is also available as a combined product with timolol maleate 0.5%. Topical CAIs are administered q 8-12 hrs. Dorzolamide (because of its lower pH) and other topical CAIs may be irritating. In rare instances, topical CAIs can cause an immune-mediated reaction that leads to severe keratitis that does not improve with topical steroids, and only resolves with discontinuance of the CAI.21 CAIs may also cause local allergic reactions, especially with long-term use. These reactions often manifest as chronic conjunctivitis with conjunctival thickening, blepharitis, mucopurulent discharge, and corneal vascularization. In mild cases, such reactions often diminish with administration of topical corticosteroids. In more severe cases, the CAI must be discontinued because of ongoing periocular inflammation. Some treated eyes tolerate a different CAI with a different vehicle.
Brinzolamide is available as a subconjunctival implant; however, studies evaluating its long-term efficacy are lacking. Further research is needed to determine if it is an effective treatment option in dogs that do not tolerate topical therapy.
Methazolamide (2-4 mg/kg PO q 8-12 hrs) is available in tablet form. Because systemic CAIs may cause vomiting, diarrhea, lethargy, metabolic acidosis with Kussmaul respiration, and hypokalemia, they have largely been replaced by topical CAIs.
Prostaglandin Analogues: Prostaglandin analogues are moderately to highly effective in dogs with acute and chronic primary glaucoma in the short and long term. They are contraindicated in the presence of anterior lens luxation and uveitis. Examples include latanoprost, bimatoprost, travoprost, and tafluprost.22,24 Prostaglandin agents are often applied q 24 hrs in the evening for maintenance therapy because diurnal pressure fluctuations are present in dogs with primary glaucoma. However, IOP is often better controlled with q 12 hrs administration because rebound mydriasis and elevated IOP may occur between dosages with once daily administration. Prostaglandin analogues induce miosis that can be extreme and interfere with vision, especially in dim light conditions. Effects of latanoprost are affected by contact lens application but not by concurrent administration of parasympatholytic drugs or prednisolone acetate.23-25
A sustained-release, intracameral implant of bimatoprost reduces IOP and episcleral venous pressure in dogs.26 Interestingly, topical prostaglandin analogue administration does not decrease episcleral venous pressure, so the intracameral device may provide better aqueous outflow and IOP control over time compared to topical therapy.
Other Therapies: Most other topical anti-glaucoma medications have been replaced by prostaglandin analogues and CAIs. Examples include selective (beta 1) and nonselective (beta 1 and beta 2) beta receptor blockers; directing-acting miotics (e.g. pilocarpine); and indirect-acting miotics (e.g. demecarium bromide). Netarsudil, a rho kinase inhibitor, has recently been studied but had no clinically-significant effects on IOP in dogs.27-29 Drug-infused implants and contact lenses have occasionally been utilized for canine glaucoma, and may represent a potential addition to topical therapy for glaucoma management.30
Surgical Therapy of Sighted Eyes
Surgical therapy is often combined with medical therapy for management of primary glaucoma. Primary glaucoma in dogs is rarely managed well long term with medical therapy alone. Certain causes/types (e.g. anterior lens luxation, melanocytic glaucoma, following intraocular surgery) of secondary glaucoma may also be treated with surgery. Various surgical techniques and interventions are available:
Cyclophotocoagulation: Laser (e.g. diode,YAG) energy is directed at the ciliary body across the sclera, or endoscopically through a corneal incision. Treatment is designed to partially destroy the ciliary body and reduce formation of aqueous humor. Laser therapy does not address drainage of aqueous humor from the eye. Laser ablation causes significant intraocular inflammation and may result in transient IOP elevation. Success of laser therapy varies. Micropulse laser therapy delivers laser energy in micropulses rather than continuously. Although it has not been shown to be more effective than traditional laser treatments, it may be an additional option to consider.31-34
Gonioimplantation: Gonioimplants are devices that divert the flow of aqueous humor into the episcleral space while bypassing the diseased iridocorneal angle. Many types are available. Historically, they have been associated with a high rate of both short- and long-term failures, primarily from occlusion and fibrosis around the endplate. Recent studies on Baervedlt® implants have reported conflicting results; however, one study found better long-term outcomes than with transscleral laser cyclophotocoagulation.35-37 Another study of 132 eyes in which an Ahmed® glaucoma valve was inserted found that vision was preserved for a mean of 56.4 months in eyes with primary glaucoma, and for 59.3 months in eyes with postoperative glaucoma.38 Eighteen eyes (13.6%) retained vision for >5 years.38 Combined laser cyclophotocoagulation and gonioimplantation may be considered for some eyes.39
Iridencleisis: Iridencleisis involves making a small scleral window and pulling a portion of the dorsal iris through the incision (Figures 13, 14). Iris is sutured to the sclera, thereby providing a pathway for flow of aqueous humor from the anterior chamber to the episcleral space. No recent studies have been published on the long-term success of iridencleisis. However, the author has used the procedure successfully to treat glaucoma unresponsive to other surgical and medical therapies. Further studies are necessary to assess the long-term results of iridencleisis.
Cryotherapy: Transscleral cyclocryotherapy may be performed to ablate the ciliary body but it tends to cause more collateral damage to surrounding ocular tissues. Cryotherapy has largely been replaced by laser cyclophotocoagulation since laser energy delivery systems more specifically target the ciliary body. While cryotherapy can potentially result in better, long-term control of IOP because of more widespread damage, hypotony, secondary phthisis bulbi, and cataract formation are possible. These complications render the eye comfortable, albeit blind. Currently, cryotherapy is not typically utilized as a vision-sparing procedure.
Lensectomy: Removal of the lens is the preferred treatment for anterior lens luxations resulting in glaucoma.
Surgical Therapy of Blind Eyes
Salvage procedures are often considered for irreversibly blind eyes, especially when IOP becomes refractory to the above treatments.
Enucleation: Enucleation, with or without intraorbital prosthesis, is a good option for alleviating pain associated with glaucoma (i.e. IOP >50 mmHg) refractory to all initiated treatments. Enucleation may also be considered for blind eyes with glaucoma that is poorly controlled on medical therapy (i.e. IOP >30 mmHg), or when medical therapy becomes too expensive or overwhelming for the owner. Certain causes of glaucoma (e.g. intraocular neoplasia) are also indications for enucleation.
Evisceration/Prosthesis: Evisceration and insertion of an intrascleral prosthesis is a cosmetic procedure that can be considered for eyes blind with primary glaucoma. It is contraindicated in the presence of intraocular neoplasia, infection, and chronic inflammation, as well as the presence of chronic corneal disease or keratoconjunctivitis sicca. While the complication rate with this procedure is low, it is higher than complication rates for enucleation.
Gentamicin Ciliary Ablation: Intravitreal gentamicin injection causes pharmacologic ablation of the ciliary body.40,41 It is considered only for eyes blind from primary glaucoma in which the eye is unresponsive to other therapies, and the owner declines enucleation or evisceration. Typically, a needle is passed through the lateral-dorsal pars plana of the ciliary body and 1 mL of vitreous is removed. The procedure is only done in chronic cases of glaucoma where the vitreous has become liquefied (approximately 30 days after onset of acute glaucoma) and can be aspirated. Gentamicin (25-40 mg) is injected into the vitreous, making sure the needle does not lacerate the lens. Dexamethasone 0.25 mg may be added to the gentamicin prior to injection.
The procedure has a less predictable outcome than either enucleation or intrascleral prosthesis; however, success rates as high as 95% (range 65-95%) are reported.40-42 Although IOP is immediately reduced to very low levels, it is possible for the ciliary body to recover in some eyes. Long term, the eye may be hypo-, normo- or hypertensive. At doses of >30 mg, gentamicin concentrations are measurable in the blood but not at levels typically ototoxic or nephrotoxic.38 However, caution must be taken in small animals so the injected amount does not exceed the recommended daily dose. Topical corticosteroids are often required for at least 1 month post-injection to control uveitis caused by the procedure.
Complications include hyphema, recurrence of glaucoma, phthisis bulbi, cataract formation, and chronic uveitis. In addition, a study evaluating enucleated eyes that had prior gentamicin injections, found 40% of them contained intraocular tumors.43 More than one-third of these tumors were malignant and exhibited aggressive behavior. No causative link was established between gentamicin injection and tumor formation, and it is possible that neoplastic tissue was present prior to the injection. These enucleated globes may represent only a small percentage of gentamicin-injected globes, so care must be taken to ensure eyes are tumor-free (e.g. via ultrasonography) before administering intravitreal gentamicin.
Cidofovir Ciliary Ablation: Cidofovir may be injected intravitreally similar to gentamicin.44 Cidofovir is an anti-viral agent typically used to treat HIV in people that was found to have ocular hypotonic effects. In one study, diluted intravitreal cidofovir and concurrent subconjunctival triamcinolone controlled IOP in 85% of treated canine eyes.42 Repeat injections (when necessary) raised the success rate to 97%.44 It should be noted; however, that 70% of injected globes became phthisical, and a small percentage developed corneal ulceration (2.4%) or endophthalmitis (2.4%).44
SUPPORTIVE THERAPY
Treatment of underlying conditions causing secondary glaucoma (e.g. uveitis, lens luxation) is indicated. For uveitis, topical and oral anti-inflammatory therapy may be indicated. Analgesics may also be helpful but oral analgesics do not alleviate pain associated with severe glaucoma, if IOP is not reduced.45 Medications that may increase IOP, such as mydriatics (e.g. atropine, cyclopentolate, tropicamide) and nonsteroidal anti-inflammatory drugs (can decrease aqueous outflow) are avoided in most eyes with glaucoma. Interestingly, mydriatic medications do not appear to negate the anti-glaucoma properties of latanoprost.24
MONITORING
It is important to note that elevated IOP can quickly cause permanent damage to the optic nerve and retina. IOP must be monitored frequently until glaucoma is controlled. Patients with significantly elevated IOPs must be reassessed within hours, not days, after therapy is instituted. Goal is to return IOP to normal (i.e. <25 mmHg, ideally 10-15 mmHg). Sustained IOPs >30 mmHg indicate the need for more aggressive therapy. If medical therapy fails, further surgical intervention should be considered. IOP can spike and not be detected by periodic monitoring of IOP, so educate the client to monitor for signs of both subtle and severe pressure elevation.
PROGNOSIS
Prognosis for primary glaucoma is guarded. Warn most owners that therapy is not curative and although exceptions exist, most affected eyes eventually go blind. Prognosis for secondary glaucoma is dependent upon the underlying cause and whether it can be treated effectively.
Preventive Measures:
Screening Tests
Genetic Test: Because genetic mutations for canine glaucoma vary widely between and within canine breeds, they currently have limited value as a screening tool for the general canine population, and are not predictive of which individuals will develop glaucoma.10 However, genetic tests can be used to identify at-risk animals and make decisions on whether they should be used for breeding.
Gonioscopy: Because goniodysgenesis is present at birth and closure of the iridocorneal angle occurs early in life, breeding animals may be screened by gonioscopy to identify at-risk dogs. Goniodysgenesis in genetically normal dogs has been linked to development of glaucoma over time and caution should be exercised when deciding whether to breed dogs with goniodysgenesis, even if IOP is normal at the time of gonioscopic examination. Gonioscopic findings; however, do not predict whether or when the eye may develop glaucoma.
IOP Measurement: Intermittent monitoring of IOP may be beneficial in some patients; however, general IOP monitoring of individuals in at-risk breeds is of no practical benefit.
Prophylactic Therapy
For dogs with primary glaucoma, prophylactic treatment of the unaffected, contralateral eye can significantly delay or prevent the onset of primary glaucoma. Even if a patient undergoes a salvage procedure for a blind, uncomfortable eye, it is imperative that glaucoma be identified as primary (via gonioscopy, histopathology, genetic testing) so that prophylactic therapy can be instituted in the contralateral eye.
Studies comparing efficacy of topical prophylactic drugs have reported mixed results regarding which medication is best. One study showed no significant differences in median time (200-300 days) to failure for demecarium bromide 0.125-0.25%, latanoprost 000.5%, and dorzolamide 2%.46 The study included eyes on concurrent anti-inflammatory (e.g. dexamethasone, prednisolone acetate) therapy, and no statistical difference was found between eyes receiving or not receiving corticosteroids.46 Results of the study suggested; however, that topical steroids may be beneficial given the inflammatory changes that were seen on histopathology of affected eyes.46
Another study showed that demecarium 0.125% administered q 24 hrs with a topical corticosteroid delayed onset of PCAG for 31 months, while betaxolol 0.25% q 12 hrs delayed onset for 30.7 months.47 A recent review of current veterinary ophthalmologists’ use of prophylactic therapy showed a variety of preferences. The important conclusion was that some form of prophylactic therapy should be used in the “normal” sighted eye after treatment or loss of the affected eye to primary glaucoma.48 Eyes on prophylactic therapy should be monitored with IOP measurements q 3-6 months to ensure that IOP is within the target range of 10-15 mmHg.
Special Considerations:
Other Resources
Recent VIN Message Board discussions on glaucoma
Recent VIN Message Board discussions on secondary glaucoma
Recent VIN Message Board discussions on primary glaucoma
Recent VIN Message Board discussions on glaucoma nonsurgical therapy
Recent VIN Message Board discussions on glaucoma surgery
Recent VIN Message Board discussions on tonometers
Proceedings articles that discuss glaucoma
Client Handout on glaucoma in dogs and cats
Medical FAQ on glaucoma therapy
2016 VIN Rounds entitled Glaucoma is not a Tonopen Number
Ophthalmology Fun Cases:
Ophthalmology Fun Case 24
Ophthalmology Fun Case 30
Ophthalmology Fun Case 131
Ophthalmology Fun Case 133
Ophthalmology Fun Case 190
Ophthalmology Fun Case 195
For more images see the Glaucoma – Dog slideshow in the Image Library
For more images see the Buphthalmos – Dog slideshow in the Image Library
Differential Diagnosis:
Exophthalmos
Lagophthalmos
Other causes of corneal edema
Episcleritis
Uveitis
References:
- Ahram D F, Cook A C, Kecova H, et al: Identification of genetic loci associated with primary angle-closure glaucoma in the basset hound. Mol Vis 2014 Vol 20 (0) pp. 497-510.
- Oliver J A, Forman O P, Pettitt L, et al: Two Independent Mutations in ADAMTS17 Are Associated with Primary Open Angle Glaucoma in the Basset Hound and Basset Fauve de Bretagne Breeds of Dog. . PLoS One 2015 Vol 10 (10) pp. e0140436.
- Kanemaki N1, Tchedre KT, Imayasu M, et al: Dogs and Humans Share a Common Susceptibility Gene SRBD1 for Glaucoma Risk. PLoS One 2013 Vol 8 (9) pp. e74372.
- Kuchtey J, Olson L M, Rinkoski T, et al: Mapping of the disease locus and identification of ADAMTS10 as a candidate gene in a canine model of primary open angle glaucoma. PLoS Genet 2011 Vol 7 (2) pp. e1001306.
- Ahonen S J, Kaukonen M, NussdorferF D, et al: A novel missense mutation in ADAMTS10 in Norwegian Elkhound primary glaucoma. PLoS One 2014 Vol 9 (11) pp. e111941.
- Sargan D R, Withers D, Pettitt L, et al: Mapping the mutation causing lens luxation in several terrier breeds. J Hered 2007 Vol 98 (5) pp. 534-8.
- Farias F H G, Johnson G S, Taylor J F, et al: An ADAMTS17 splice donor site mutation in dogs with primary lens luxation. Invest Ophthalmol Vis Sci 2010 Vol 51 (9) pp. 4716-21.
- Gould D,Pettitt L, McLaughlin B, et al: ADAMTS17 mutation associated with primary lens luxation is widespread among breeds.. Vet Ophthalmol 2011 Vol 14 (6) pp. 378-84.
- Peterson-Jones SM, Forcier J, Mentzer AL: Ocular melanosis in the Cairn Terrier: clinical description and investigation of mode of inheritance . Vet Ophthalmol 2007 Vol 10 (Suppl 1) pp. 63-69.
- Graham K L, McCowan C, White A: Genetic and Biochemical Biomarkers in Canine Glaucoma.. Vet Pathol 2017 Vol 54 (2) pp. 194-203.
- Grozdanic S D, Kecova H, Harper M M, et al: Functional and structural changes in a canine model of hereditary primary angle-closure glaucoma. Invest Ophthalmol Vis Sci 2015 Vol 51 (1) pp. 255-63.
- Ae Park S, Sledge D, Monahan C, et al : Primary angle-closure glaucoma with goniodysgenesis in a Beagle dog. BMC Vet Res Marc Vol 15 (1) pp. 75.
- Kato K, Sasaki N, Matsunaga S, et al : Possible association of glaucoma with pectinate ligament dysplasia and narrowing of the iridocorneal angle in Shiba Inu dogs in Japan. Vet Ophthalmol 2006 Vol 9 (2) pp. 71-5.
- Bjerkas E, Ekesten B, Farstad W: Pectinate ligament dysplasia and narrowing of the iridocorneal angle associated with glaucoma in the English Springer Spaniel. Vet Ophthalmol. 2002 Vol 5 (1) pp. 49-54.
- Ekesten B, Narfström K: Correlation of morphologic features of the iridocorneal angle to intraocular pressure in Samoyeds. Am J Vet Res 1991 Vol 52 (11) pp. 1875-8.
- Miller P E, Bentley E: Clinical Signs and Diagnosis of the Canine Primary Glaucomas. Vet Clin North Am Small Anim Pract, VI ed. 2015 Vol 45 (6) pp. 1183-212.
- Gelatt, KN: Prevalence of the breed-related glaucomas in purebred dogs in North America, in Proceedings. 34th Annual Meeting of the American College of Veterinary Ophthalmolists 2003 pp. 73.
- Herring IA, Morgan RV : Glaucoma. Handbook of Small Animal Practice, 5 ed. Elsevier Saunders, St. Louis 2008 pp. 979-85.
- Fricker G, Smith K, Gould D J: Survey of the incidence of pectinate ligament dysplasia and glaucoma in the UK Leonberger population. Vet Ophthalmol 2016 Vol 19 (5) pp. 379-85.
- Bedford P G C: Open-angle glaucoma in the Petit Basset Griffon Vendeen. Vet Ophthalmol 2017 Vol 20 (2) pp. 98-102.
- Beckwith-Cohen B, Bentley E, Gasper D J, et al: Keratitis in six dogs after topical treatment with carbonic anhydrase inhibitors for glaucoma. J Am Vet Med Assoc 2015 Vol 247 (12) pp. 1419-26.
- Kwak J, Kang S, Lee E-R, et al: Effect of preservative-free tafluprost on intraocular pressure, pupil diameter, and anterior segment structures in normal canine eyes.. Vet Ophthalmol 2017 Vol 20 (1) pp. 34-39.
- Kahane N, Bdolah-Abram T, Raskansky H, et al: The effects of 1% prednisolone acetate on pupil diameter and intraocular pressure in healthy dogs treated with 0.005% latanopros. Vet Ophthalmol 2016 Vol 19 (6) pp. 473-79.
- Kahane N, Raskansky H, Bdolah-Abram T, et al: The effects of topical parasympatholytic drugs on pupil diameter and intraocular pressure in healthy dogs treated with 0.005% latanoprost. . Vet Ophthalmol 2016 Vol 19 (6) pp. 464-72.
- Hatzav M, Bdolah-Abram T, Ofri R: Interaction with therapeutic soft contact lenses affects the intraocular efficacy of tropicamide and latanoprost in dogs. J Vet Pharmacol Ther 2016 Vol 39 (2) pp. 138-43.
- Lee SS, Burke J, Shen J, et al: Bimatoprost sustained-release intracameral implant reduces episcleral venous pressure in dogs. Vet Ophthalmol 2018 Vol 21 (4) pp. 376-81.
- Leary K A, Steibel J P, Harman C D, et al: Safety and efficacy of topically administered netarsudil-latanoprost fixed dose combination (FDC; Rocklatan™) in normal and glaucomatous dogs with ADAMTS10-open-angle glaucoma (ADAMTS10-OAG).. 2021 Vol 24 (6) pp. 610-19.
- Leary K A, Steibel J P, Harman C D, et al: Safety and efficacy of topically administered netarsudil (Rhopressa™) in normal and glaucomatous dogs with ADAMTS10-open-angle glaucoma (ADAMTS10-OAG).. Vet Ophthalmol 2021 Vol 24 (Supple1 (0)) pp. 75-86.
- Yang V Y, Miller P E, Keys D A, et al: Effects of 0.02% netarsudil ophthalmic solution on intraocular pressure of normotensive dogs.. Vet Ophthalmol 2021 Vol 24 (Supple 1(0)) pp. 87-95.
- Miller P E, Eaton J S: Medical anti-glaucoma therapy: Beyond the drop.. Vet Ophthalmol 2021 Vol 24 (Supple 1(0)) pp. 2-15.
- Sebbag L, Allbaugh R A, Strauss R A, et al: MicroPulse™ transscleral cyclophotocoagulation in the treatment of canine glaucoma: Preliminary results (12 dogs). Vet Ophthalmol 2019 Vol 22 (4) pp. 407-414.
- Sebbag L, Crabtree E E, Sapienza J S, et al : Corneal hypoesthesia, aqueous tear deficiency, and neurotrophic keratopathy following micropulse transscleral cyclophotocoagulation in dogs. Vet Ophthalmol 2020 Vol 23 (1) pp. 171-80.
- Sapienza JS, Kim K, Rodriguez E, et al: Preliminary findings in 30 dogs treated with micropulse transscleral cyclophotocoagulation for refractory glaucoma.. Vet Ophthalmol 2019 Vol 22 (4) pp. 520-58.
- Story B D, Sapienza J S, Di Girolamo N, et al : Long-term results (>1 year) in 19 dogs treated with MicroPulse transscleral diode cyclophotocoagulation for refractory glaucoma. Vet Ophthamol 2021 Vol 24 (6) pp. 572-81.
- Crowe Y C, Groth A D, White J, et al : Outcomes of Baervedlt implant surgery in 17 dogs (20 eyes) with primary closed-angle glaucoma (2013–2019). . Vet Ophthalmol 2021 Vol 24 (supple 1(0)) pp. 109-115.
- Crowe Y C, Groth A D, White J, et al : Outcomes of Baervedlt implant surgery in 17 dogs (20 eyes) with primary closed-angle glaucoma (2013–2019). . Vet Ophthalmol 2021 Vol 24 (supple 1(0)) pp. 109-115.
- Graham K L, Hall E J S, Caraguel C, et al : Comparison of diode laser trans-scleral cyclophotocoagulation versus implantation of a 350-mm2 Baerveldt glaucoma drainage device for the treatment of glaucoma in dogs (a retrospective study: 2010-2016).. Vet Ophthalmol 2018 Vol 21 (5) pp. 487-97.
- Saito A, Iwashita H, Kazama Y, et al: Long-term vision outcomes and breed differences of Ahmed Glaucoma Valve implantation in 132 eyes of 122 dogs. Vet Ophthalmol 2021 Vol 25 (2) pp. 118-27.
- Sapienza J S, van der Woerdt A: Combined transscleral diode laser cyclophotocoagulation and Ahmed gonioimplantation in dogs with primary glaucoma: 51 cases (1996-2004. Vet Ophthalmol 2005 Vol 8 (2) pp. 121-27.
- Rankin A J, Lanuza R L, KuKanich B, et al: Measurement of plasma gentamicin concentrations postchemical ciliary body ablation in dogs with chronic glaucoma. Vet Ophthalmol 2016 Vol 19 (1) pp. 57-62.
- Bingaman DP, Lindley DM, Glickman NW et al: Intraocular Gentamicin and Glaucoma: A Retrospective Study of 60 Dog and Cat Eyes (1985 – 1993). Vet Comp Ophthalmol 1994 Vol 4 pp. 113.
- Julien ME, Schechtmann SA, Michau TM, et al: Pharmacologic ciliary body ablation for chronic glaucoma in dogs: A retrospective review of 108 eyes from 2013 to 2018. Vet Ophthalmol 2021 Vol 24 (Suppl 1) pp. 125-30.
- Duke F D, Strong T D, Bentley E, et al: Canine ocular tumors following ciliary body ablation with intravitreal gentamicin. Vet Ophthalmol 2013 Vol 16 (2) pp. 159-62.
- Low M C, Landis M L, Peiffer R L: Intravitreal cidofovir injection for the management of chronic glaucoma in dogs. Vet Ophthalmol 2014 Vol 17 (3) pp. 201-06.
- Zibura A E, Salmon J H, Lopez B B, et al : Glaucoma-associated pain results in mechanical sensitivity changes in dogs: A pilot study. Vet Ophthalmol 2021 Vol 24 (Suppl 1 (0)) pp. 116-24.
- Dees DD, Fritz KJ, Maclaren NE, et al: Efficacy of prophylactic antiglaucoma and anti-inflammatory medications in canine primary angle-closure glaucoma: a multicenter retrospective study (2004-2012). Vet Ophthalmol 2014 Vol 17 (3) pp. 195-200.
- Miller P E, Schmidt G M, Vainisi S J, et al : The efficacy of topical prophylactic antiglaucoma therapy in primary closed angle glaucoma in dogs: a multicenter clinical trial. J Am Anim Hosp Assoc 2000 Vol 36 (5) pp. 431-8.
- Plummer C E, Bras D, Grozdanić S, et al : Prophylactic anti-glaucoma therapy in dogs with primary glaucoma: A practitioner survey of current medical protocols. . Vet Ophthalmol 2021 Vol 24 (Suppl 1 (0)) pp. 96-108.
Feedback:
If you note any error or omission or if you know of any new information, please send your feedback to VINcyclopedia@vin.com.
If you have any questions about a specific case or about this disease, please post your inquiry to the appropriate message boards on VIN.