Establishing a reproducible method for the culture of primary equine corneal cells
Rachel L. Davis1,2 | Ursula M. Dietrich2 | Thomas M. Krunkosky3 | David J. Hurley4 | Adrian J. Reber4
Abstract
Objective
To establish a reproducible method for the culture of primary equine corneal epithelial cells, keratocytes, and endothelial cells and to describe each cell’s morphologic characteristics, immunocytochemical staining properties and conditions required for cryopreservation.
Procedures
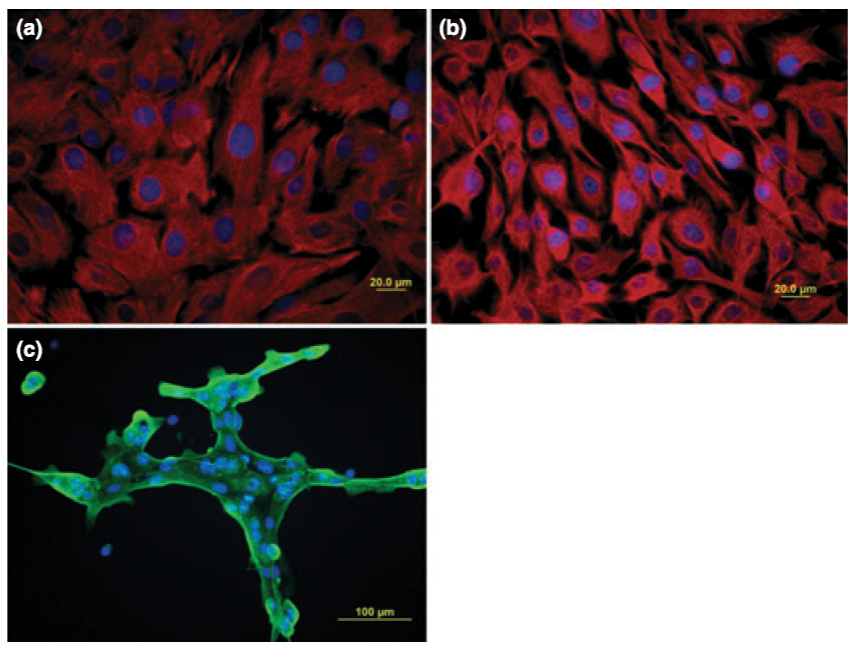
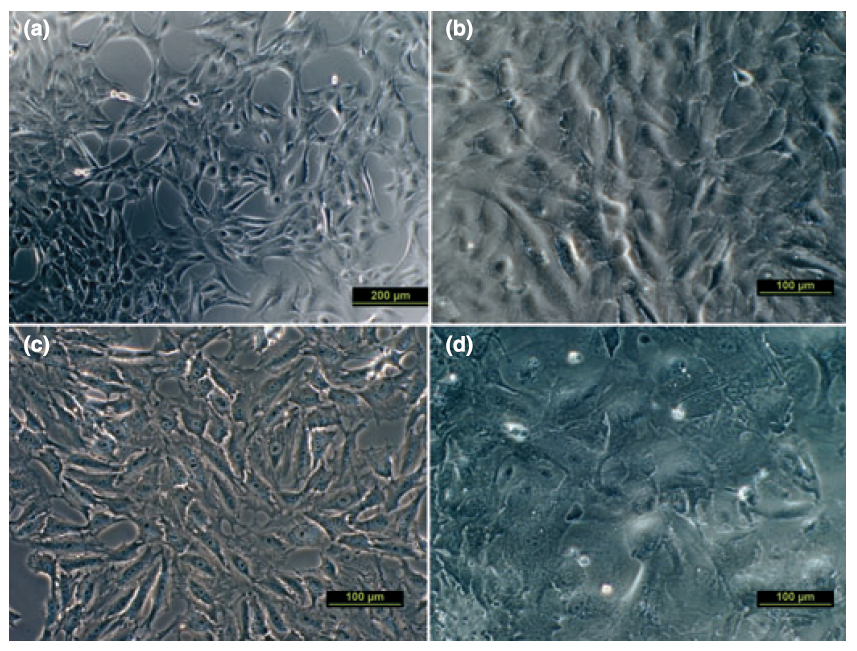
All three equine corneal cell types were successfully grown in culture. Cultured corneal endothelial cells were large, hexagonal cells with a moderate growth rate. Keratocytes were small, spindloid cells that grew rapidly. Epithelial cells had heterogenous morphology and grew slowly. The endothelial cells and keratocytes stained positive for vimentin and were morphologically distinguishable from one another. The epithelial cells stained positive for cytokeratin. Keratocytes and endothelial cells were able to be cryopreserved and recovered. The cryopreserved cells maintained their morphological and immunocytochemical features after cryopreservation and recovery.
Results
This work establishes reproducible methods for isolation and culture of equine corneal keratocytes and endothelial cells. Cell morphology and cytoskeletal element expression for equine corneal epithelial cells, keratocytes, and endothelial cells are also described. This has not previously been reported for equine corneal cells. This report also demonstrates the ability to preserve equine keratocytes and endothelial cells for extended periods of time and utilize them long after the primary-cell collection, a feature that has not been reported for veterinary corneal cell culture.
Discussion
Intravitreal dexamethasone injections in dogs with ocular blastomycosis is a simple procedure that significantly reduced the need for enucleation of eyes affected with ocular blastomycosis. This procedure may help prevent loss of eyes in dogs affected with ocular blastomycosis. None.
Images
Keywords: equine corneal cell culture, horse corneal epithelial cells, equine keratocyte culture, equine corneal endothelial cells, veterinary ophthalmology corneal research, cryopreservation equine corneal cells, immunocytochemical staining horse cornea, primary corneal cell isolation horses, equine corneal morphology study, veterinary corneal cell preservation
1Animal Eye Clinic, Westfield, Indiana, USA
2Department of Small Animal Medicine and Surgery, College of Veterinary Medicine, University of Georgia, Athens, GA USA
3Department of Anatomy and Radiology, College of Veterinary Medicine, University of Georgia, Athens, GA 30602, USA
5Department of Population Health, College of
Veterinary Medicine, University of Georgia, Athens, GA 30602, USA
Correspondence:
Rachel L. Davis, DVM, MS, Diplomate, ACVO – Ophthalmologist
Animal Eye Clinic
4750 Killarney Drive
Carmel, IN 46033
Email: info@indyaec.com