Leiomyoma of the third eyelid in a dog
Rachel L. (Mathes) Davis1 | Sarah J. Noble2 | Angela E. Ellis3
Abstract
A 14-year-old neutered male Dachshund presented for the evaluation of oculus dexter (OD) third eyelid elevation ongoing for approximately 2 months. Complete ophthalmic examination revealed a large, nonpainful, well-demarcated, soft mass at the base of the right third eyelid causing elevation and mild hyperemia. The mass was freely moveable with the third eyelid, and no right globe deviation was noted. No other abnormalities were noted on physical examination, routine blood chemistry, complete blood count, serum T4, urinalysis, or urine cortisol/creatinine ratio. Ocular B-mode ultrasonography showed an anechoic, well-demarcated, homogenous, soft tissue mass at the base of the third eyelid with no orbital extension. A leiomyoma was diagnosed after multiple punch biopsies were obtained from the palpebral surface of the mass. The right third eyelid was excised surgically. Histopathology confirmed a completely excised, nodular, unencapsulated, expansile mass within the third eyelid. Positive smooth muscle actin and negative S-100 immunohistochemistry confirmed a leiomyoma. Bundles of normal smooth muscle were also present adjacent to the mass. The mass was compressing the adjacent lacrimal gland and associated with moderate dacryoadenitis. Twelve months postoperatively, the right globe position and motility remain normal with no evidence of mass regrowth. To the author’s knowledge, this is the first reported case of a leiomyoma of the third eyelid in any species. In this case, the mass was completely excised and no regrowth has occurred twelve months after surgery. This case along with independently reviewed canine third eyelids clearly demonstrates the presence of smooth muscle within the canine third eyelid.
Keywords: canine, eyelid leiomyoma, leiomyoma, smooth muscle, third eyelid
1Animal Eye Clinic, Westfield, Indiana, USA
2Portland Veterinary Specialists, Portland, ME, USA
3College of Veterinary Medicine, Athens Veterinary Diagnostic Laboratory, University of Georgia, Athens, GA, USA
Correspondence:
Rachel L. Davis, DVM, MS, Diplomate, ACVO – Ophthalmologist
Animal Eye Clinic
4750 Killarney Drive
Carmel, IN 46033
info@indyaec.com
INTRODUCTION
Canine leiomyomas are benign tumors arising from smooth muscle and are most commonly found in the gastrointestinal tract.1–7 These tumors may also occur in a
variety of other locations including the heart,8 urinary system, 9–12 uterus,13 mesosalpinx,14 vagina,15,16 trachea,17 and nasopharynx.18 Angioleiomyomas, uncommonly reported in dogs, arise from vascular smooth muscle and may occur in a variety of locations. Although leiomyomas are benign, they may cause serious disease secondary to compression or displacement of adjacent abdominal or alimentary structures.1,2,5,6 Leiomyomas may also produce insulin-like growth factors that cause hypoglycemia in affected canine patients.19–21 In spite of their relative commonness in other locations in dogs, there is only one report of a canine intraocular leiomyoma, arising from the iris in a dog.22 In the human literature, there are multiple reports of ocular or periocular leiomyomas arising from the orbit,23 eyelid,24,25 conjunctiva,26 iris,27–29 ciliary body,30–33 and choroid.34,35 Ocular leiomyomas are considered rare36,37 as these case reports are very sporadic throughout the literature. Additionally, a 1994 study revealed that many earlier intraocular ‘leiomyomas’ were misdiagnosed; immunohistochemistry on previous cases confirmed many of these tumors to be melanocytic lesions.38 Thus, ocular leiomyomas may be even rarer than the literature would suggest. Mesectodermal leiomyomas are benign intraocular tumors arising from the ciliary body epithelium, a cell of neural crest origin.39 Although routine histopathology may be highly suggestive of a leiomyoma, a definitive diagnosis of leiomyoma can only be made with immunohistochemistry, as the clinical and microscopic appearance of these tumors may be similar to other tumors such as melanocytic neoplasia, gastrointestinal stromal tumors (GISTs), and other spindle cell tumors.2,3,38 This was also highlighted in a 2007 study of canine small intestinal and cecal tumors that demonstrated distinct immunohistochemical differences between gastrointestinal smooth muscle and stromal tumors, resulting in many tumors being reclassified.40
The herein present case report describes a primary canine third eyelid leiomyoma diagnosed with histopathology and immunohistochemistry. To the authors’ knowledge, this is the first report of a primary third eyelid leiomyoma in any species. The mass was surgically excised with no regrowth reported twelve months postoperatively. This case report also definitively demonstrates the presence of native smooth muscle within the canine third eyelid, the presence of which has previously been indeterminate.
CLINICAL REPORT
Ophthalmic examination
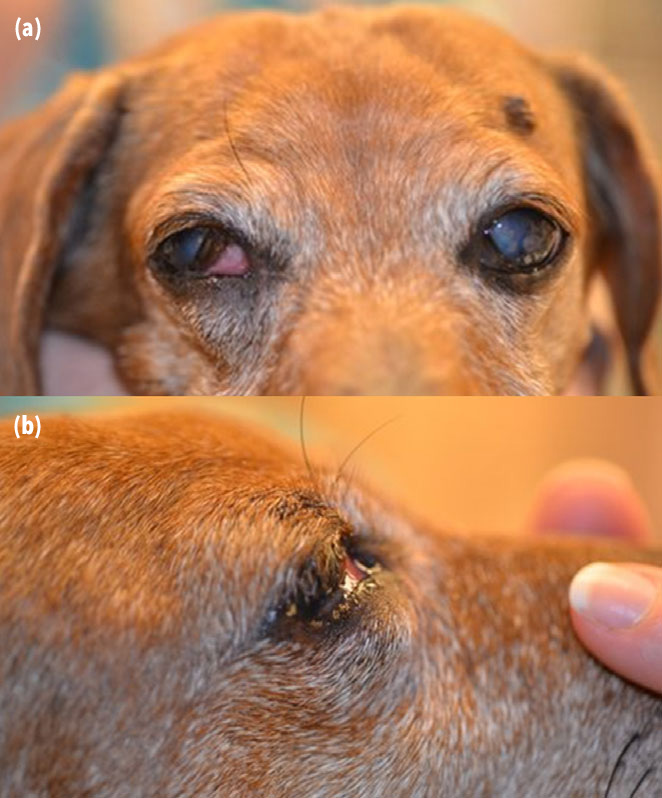
A fourteen-year-old neutered male short-haired Dachshund presented to a private referral hospital’s Ophthalmology Service for the evaluation of recurrent corneal ulceration oculus uterque (OU), blepharospasm OU, and white corneal opacities OU. Full ophthalmic examination with slit-lamp biomicroscopy (Kowa SL-15; Kowa Optimed, Torrance, CA, USA) and indirect ophthalmoscopy (Keeler Vantage Plus; Dan Scott and Associates, Westerville, OH, USA) performed by a board-certified veterinary ophthalmologist (RM) revealed mild enophthalmos OU, mild intermittent blepharospasm OU, mild-to-moderate hyperemia OU, paraxial ventrotemporal dense mid to anterior corneal stromal lipid opacifications OU and multifocal wispy arborizing anterior stromal limbal-based corneal vascularization temporally and ventrotemporally extending variable distances into the corneal stroma OU. A large, nonpainful, well-demarcated, soft mass at the base of the right third eyelid causing third eyelid elevation was also identified (Fig. 1). Save for the previously mentioned mild OU enophthalmos, there was no OD globe deviationor strabismus. There was normal ocular motility and an otherwise normal neuro-ophthalmic examination. The mass was freely movable within the right third eyelid. Schirmer tear test values were 18 mm/min OD and 15 mm/min OS (Schirmer Tear Test; Merck and Company, Inc., Whitehouse Station, NJ, USA), and intraocular pressures were 8 mmHg OD and 8 mmHg OS measured by applanation tonometry (Tonopen XL; Reichert Technologies, Depew, NY, USA). The tear film breakup time (TBUT), measured as previously described,41 was decreased in both eyes (3 s OD, 5 s OS), consistent with a qualitative tear deficiency. There was no corneal or conjunctival fluorescein stain retention OU. The patient was visual with a normal intraocular examination OU. Based on the ophthalmic examination, diagnoses of OU qualitative tear deficiency, OU corneal lipidosis, OU mild keratitis, and OD third eyelid mass were made. Recommendations were made for systemic workup to identify any underlying systemic factors contributing to the corneal lipidosis along with an ocular ultrasound and biopsies of the third eyelid mass. Topical therapy was instituted with 0.2% cyclosporine (small strip OU BID; Optimmune, Merck, White House Station, NJ, USA) and an artificial lacrimomimetic (1 drop OU BID; Refresh Celluvisc, Allergan, Irvine, CA, USA) for the qualitative tear deficiency.
Diagnostics
Systemic diagnostics including routine blood chemistry, complete blood count, serum triglycerides, serum T4, and urinalysis were within normal limits. A urine cortisol/creatinine ratio was 6, which is consistent with normal adrenal function. Based on these diagnostics, no further investigation of adrenal or thyroid function was performed.
The patient presented again for ocular ultrasound and biopsies of the third eyelid mass. Ophthalmic examination revealed marked improvement in the patient’s overall comfort. The hyperemia and enophthalmos had resolved OU. The limbal-based corneal vascularization had resolved, and the tear film breakup time (TBUT) was normal in both eyes (>15 s OU). No other changes in the ophthalmic examination were noted. The right third eyelid mass appeared unchanged.
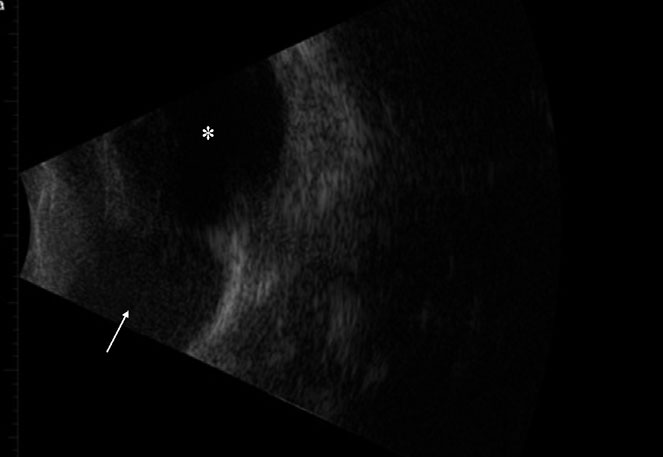
The patient was sedated with dexmedetomidine (375 mcg/m² IV; Dexdomitor, Zoetis, Florham Park, NJ, USA) and butorphanol (0.4 mg/kg IV; Torbugesic, Fort Dodge, Fort Dodge, IA, USA). After sedation, ocular ultrasound imaging (10 mHz UBM probe; Aviso, Quantell Medical, Bozeman, MT, USA) was obtained and showed an anechoic to mildly hypo-echoic, well-demarcated, homogenous, soft tissue mass at the base of the third eyelid with no orbital extension (Fig. 2). Based on the ultrasound and readily accessible location for tissue sampling, biopsies of the mass were pursued. After routine surgical preparation, local anesthetic was injected subconjunctivally on the palpebral surface of the third eyelid overlying the mass with 2% injectable lidocaine (0.15 mL subconjunctival; MWI, Boise, ID, USA). Several 4-mm punch biopsies (disposable biopsy punch; Miltex, York, PA, USA) were obtained from the palpebral surface of the third eyelid in the body of the firm, intrapalpebral mass and submitted for histopathologic examination. The conjunctival defect was closed with simple interrupted 6-0 vicryl (Ethicon; Johnson and Johnson, New Brunswick, NJ, USA) sutures. The patient’s dexmedetomidine sedation was then reversed using atipamezole (375mcg/m² IM; Antisedan, Zoetis, Florham Park, NJ, USA). The patient recovered routinely and was discharged with topical diclofenac (1 drop Q12 h OD; Nexus Pharmaceuticals, Vernon Hills, IL, USA) and topical Optimmune as previously prescribed.
Third eyelid biopsy histopathology
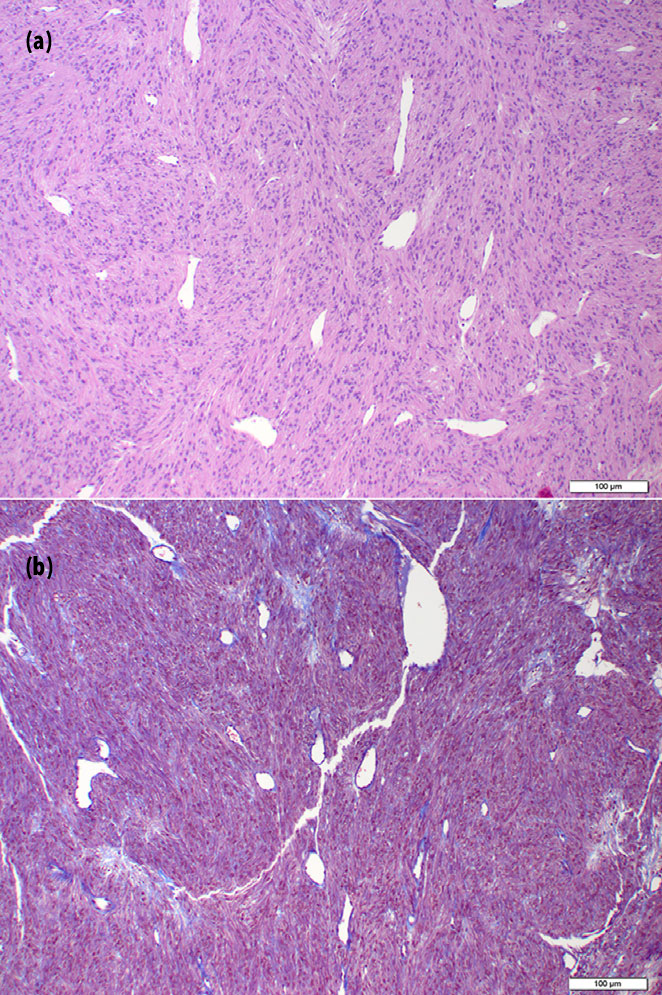
Examined sections contained sections of a mass that were composed of interlacing bundles and fascicles of spindle shaped to polygonal cells with indistinct borders and scant, clear to eosinophilic cytoplasm that extended away from the nucleus in delicate streams (Fig. 3). Nuclei were round to oval with finely stippled chromatin and 1-2 distinct nucleoli. Anisocytosis and anisokaryosis were mild. The mass also had numerous, mildly dilated, occasionally branching vessels lined by flattened to slightly plump endothelial cells. A Masson’s trichrome stain was performed, and staining was consistent with muscle (Fig. 3). A diagnosis of leiomyoma was made; however, an angioleiomyoma could not be excluded.
Right third eyelid excision
Based on the histopathology results and location of the mass, recommendations were made for surgical excision of the right third eyelid with histopathologic evaluation. The patient was premedicated with acepromazine (0.02 mg/kg IM; Fort Dodge, Fort Dodge, IA, USA) and hydromorphone (0.1 mg/kg IM; West Ward, Eatontown, NJ, USA) prior to general anesthesia. Anesthetic induction was achieved with propofol (4 mg/kg IV; Propoflo, Abbott Animal Health, North Chicago, IL, USA) to effect. General anesthesia was maintained after endotracheal intubation with inhalant isoflurane 1–2% in oxygen (Isoflo; Abbott Animal Health, North Chicago, IL, USA). After endotracheal intubation and anesthesia maintenance, cefazolin (22 mg/kg IV; WG Critical Care LLC, Paramus, NJ, USA) and carprofen (2.2 mg/kg SQ; Rimadyl, Zoetis, Florham Park, NJ, USA) were then administered. The patient was positioned in sternal recumbency, and the right eye was prepped routinely for surgery (Fig. 4). Prior to excision, an encircling conjunctival incision was made at the base of the third eyelid with a #15 blade. Metzenbaum scissors were used to undermine the bulbar and palpebral conjunctiva on either side of the third eyelid within the fornices to provide a section of conjunctiva available for surgical closure after third eyelid excision. The right third eyelid was excised at the base as previously described⁴² and submitted for histopathology. The conjunctiva was closed with simple continuous 6-0 vicryl (Ethicon; Johnson and Johnson, New Brunswick, NJ, USA) sutures using with care to place the knots deep within the dorsonasal and ventrotemporal fornices on either end of the incision to avoid contact with the cornea.

The patient recovered from general anesthesia routinely. Postoperatively, the patient received hydromorphone (0.05 mg/kg IV Q6 h). The patient was discharged with topical OD diclofenac (1 drop OD Q8 h), Optimmune (small strip OU Q12 h) neomycinpolymyxin gramicidin (1 drop OD Q8 h; Neopolygram, Bausch and Lomb, Inc., Tampa, FL, USA), and artificial tear ointment (small strip OD Q6 h; Puralube, Fera Pharmaceuticals, Locust Valley, NY, USA) along with oral tramadol (2 mg/kg PO for 5 days; Amneal Pharmaceuticals, Glasgow, KY, USA) and carprofen (2.2 mg/kg PO Q12 h; Rimadyl, Zoetis, Florham Park, NJ, USA).
Right third eyelid and mass histopathology
Examined sections of third eyelid contained a well-demarcated, unencapsulated, nodular, expansile mass at the base of the third eyelid that compressed the adjacent third eyelid lacrimal gland (Fig. 5). The mass was composed of dense bundles of polygonal to spindle-shaped cells with indistinct borders, scant to moderate, eosinophilic to pale eosinophilic, homogeneous to finely vacuolated cytoplasm, and round, oval, or cigar shaped, lightly hyperchromatic nuclei with 1–2 distinct nucleoli. Anisocytosis and anisokaryosis were mild to moderate, and there were two mitotic figures in 10 high-powered fields of view. In some areas, neoplastic cells were separated by numerous round to oval vacuoles containing mucinous material, and there were few, small, scattered foci of mineralization. In the adjacent third eyelid lacrimal gland, ducts and acini were often ectatic with intraluminal neutrophils, macrophages, and debris, and the interstitium was mildly expanded by lymphocytes and plasma cells. Immunohistochemical staining was performed using a-smooth muscle actin and a-S100 antibodies. Neoplastic cells were diffusely and strongly positive for smooth muscle actin (Fig. 5) but were diffusely negative for S-100 (Fig. 5).
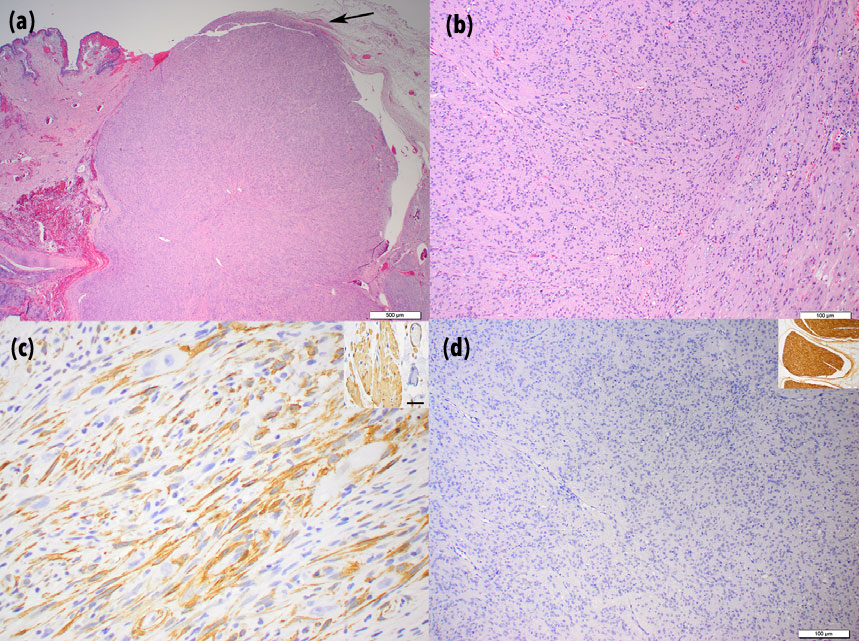
third eyelid mass are depicted. The mass is well
demarcated and is composed of densely packed,
interlacing bundles of spindle-shaped cells. A
small bundle of normal smooth muscle is evident
at the base of the tumor (arrow) H&E stain (a,
29 magnification). Neoplastic cells are spindle
shaped and are arranged in relatively well organized
bundles and fascicles. Anisocytosis and
anisokaryosis are mild, and mitoses are rare.
Nuclei are round, oval, or cigar shaped and
lightly hyperchromatic. Occasional cells have
clear or lightly basophilic vacuoles H&E stain (b,
109 magnification). Immunohistochemistry using
anti-smooth muscle actin antibody (c, 409
magnification). Neoplastic cells exhibit strong
positive staining Inset: positive control
Immunohistochemistry using anti-S100 antibody
(d, 109 magnification). Neoplastic cells are
diffusely negative for S100 Inset: Positive control.
Clinical outcome and results
The patient presented for evaluation 12 days postoperatively. The conjunctival incision was healed. The suture knots were visible in the fornices and causing mild conjunctival hyperemia. These were thus removed along with the conjunctival suture using topical proparacaine (Alcaine; Alcon Laboratories, Fort Worth, TX, USA). The ophthalmic examination was otherwise unremarkable,excepting the previously described small lipid corneal opacities. The Schirmer tear test values were 24 mm/minOD and 17 mm/min OS (Schirmer Tear Test; Merck and Company, Inc., Whitehouse Station, NJ, USA), and intraocular pressures were 14 mmHg OD and 13 mmHgOS measured by applanation tonometry (Tonopen XL; Reichert Technologies, Depew, NY, USA). Recommendations were made for long-term topical Optimmune (small strip OU Q24 h). Artificial tear ointment OD was recommended for 2 weeks with instructions to discontinue after that. All other medications were discontinued.The patient presented for evaluation 3 months after surgery (Fig. 6).

The right conjunctival surfaces were normal with the absence of the right third eyelid. Except for the previously described corneal lipidosis, the ophthalmic examination was normal. Schirmer tear test values were17 mm/min OD and 22 mm/min OS (Schirmer TearTest; Merck and Company, Inc., Whitehouse Station, NJ,USA). No corneal ulceration or episodes of discomfort had occurred since surgery.Telephone conversation with the owners 12 months after surgery revealed that there had been no change in the patient’s general or ocular condition or appearance.No clinical signs of keratoconjunctivitis sicca had been noted as the owners described bright, clear, comfortable eyes in the patient. The owners had continued the Optimmunetherapy OU Q24 h and were still administering the medication.
samples H&E stain.
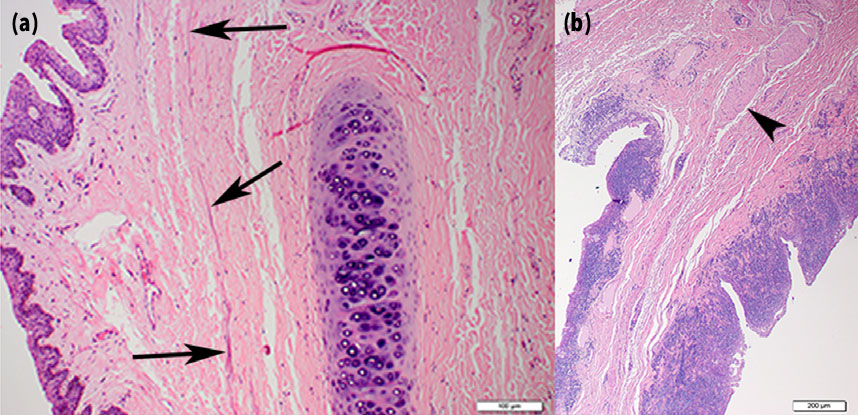
Histology of selected canine third eyelids.
Several sections of separate canine third eyelids were examined histologically to evaluate for the presence of native smooth muscle. Strips of normal smooth muscle were consistently noted within the canine third eyelid.There were variations among patients and sections as to the amount of smooth muscle present (Fig. 7), but it was consistently noted on canine third eyelid histology.
DISCUSSION
The described case herein is the first reported leiomyoma of the third eyelid in any species, to the authors’ knowledge.This tumor was diagnosed based on histopathology and immunohistochemistry. As has been previously documented,3,38,40 it is very important to perform immunohistochemistry on suspected leiomyomas as these tumors may histologically appear similar to other tumors. In our case,the tumor displayed clear histologic characteristics of mesenchymal origin. The tumor exhibited polygonal to spindle cell shape, low mitotic index, scant intercellular connective tissue matrix, and an intermittent ‘whorling’ cell pattern, which may be histologic features of other types of tumors. Because this tumor was not located within the gastrointestinal tract, a gastrointestinal stromal tumor, which may appear similar to a leiomyoma, was not considered. Peripheral nerve sheath tumors and melanocytic neoplasia have been reported within the globe and in the facial region in dogs;43–46 therefore, these tumors were considered important differential diagnoses. In this case, smooth muscle actin (SMA) staining was positive and S-100 staining was negative, clearly differentiating the tumor as a leiomyoma. The initial biopsy samples showed frequent, prominent, often dilated blood vessels within the tumor; thus, an angioleiomyoma was initially considered. However, once the tumor was available in its entirety for review, an angioleiomyoma was ruled out based on a histologic lack of similarly prominent vasculature, and a leiomyoma was diagnosed.
Leiomyomas are benign tumors and, as such, do not metastasize, but may be locally expansive and can become very large. In dogs, these tumors are most commonly found in the GI tract and are one of the most common GI tumors.2–4,40,47 In the GI tract, these tumors cause morbidity related to adjacent structure involvement; effects are usually due to compression or obstruction. In our case, the leiomyoma caused third eyelid elevation, but was found primarily as an incidental finding. This patient presented to the ophthalmologist for clinical signs of his qualitative tear deficiency. By the time the mass was removed, it had grown to a large enough size that the facial appearance of the patient was altered and the third eyelid was significantly elevated (Fig. 4). Likely, had themes continued to grow and this been the only clinical ocular abnormality, the patient would have been presented to the ophthalmologist for these changes. Because themes was limited to the third eyelid, removal of the third eyelid in its entirety was warranted. In retrospect, excision of the mass without removal of the third eyelid could has been attempted. However, this mass was quite extensive and it is unlikely that both complete excision of the mass and preservation of the third eyelid anatomy would have been achieved. A primary mass of the third eyelid is one of the rare indications for complete removal of this structures removal of the associated lacrimal gland is considered detrimental.48 In our case, close monitoring of the quantitative tear production was considered important during follow-up. In addition, the possibility of clinical keratoconjunctivitis sicca was discussed with the client prior to surgical removal of the third eyelid. The patient’s quantitative and qualitative tear production remains normal 12 months after surgery on once-per-day lacrimostimulant therapy. Whether the quantitative tear production would have been affected had this medication not been administered is unknown. In this patient’s case, this medication was administered to treat a qualitative tear deficiency, for which the patient was already clinical prior to removal of the lacrimal gland.
This case is interesting in that it is the first reported case of leiomyoma in the periocular region of a dog, but also because it clearly demonstrates the presence of native smooth muscle within the canine third eyelid. Not only does this tumor originate from smooth muscle and by default shows the presence of smooth muscle in this location, and there were also bundles of normal smooth muscle at the base of the mass within our patient’s third eyelid (Fig. 5). This prompted the authors to critically examine other canine third eyelids. A random selection of normal canine third eyelids or third eyelids submitted for other conditions clearly demonstrated the presence of smooth muscle (Fig. 7). There were variations among patients and sections as to the amount of smooth muscle present (Fig. 7), but it was consistently noted on canine third eyelid histology and was exclusively at or near the base of the third eyelid along the margins of, and usually parallel to, the gland. Previously, the presence of native smooth muscle within the canine eyelid was undecided, being more commonly reported as a consistent feature of the feline third eyelid. Further research is needed to elucidate the full role of this muscle in the dog. Leiomyoma may occur within the canine third eyelid. In our case, surgical excision carried an excellent prognosis with no tumor recurrence to date 12 months after surgery. This report also definitively illustrates the presence of native smooth muscle within the canine third eyelid. Further work is needed to determine the function of the smooth muscle in this location. Serial evaluation of the quantitative tear production in any patient after third eyelid excision is warranted.
REFERENCES
- Danova NA, Robles-Emanuelli JC, Bjorling DE. Surgical excision of primary canine rectal tumors by an anal approach in twenty-three dogs. Veterinary Surgery 2006; 35: 337–340.
- Frost D, Lasota J, Miettinen M. Gastrointestinal stromal tumors and leiomyomas in the dog: a histopathologic, immunohistochemical, and molecular genetic study of 50 cases. Veterinary Pathology 2003; 40: 42–54.
- Gillespie V, Baer K, Farrelly J et al. Canine gastrointestinal stromal tumors: immunohistochemical expression of CD34 and examination of prognostic indicators including proliferation markers Ki67 and AgNOR. Veterinary Pathology 2011; 48: 283–291.
- Willard MD. Alimentary neoplasia in geriatric dogs and cats. The Veterinary Clinics of North America. Small Animal Practice 2012; 42: 693–706, vi.
- Beck JA, Simpson DS. Surgical treatment of gastric leiomyoma in a dog. Australian Veterinary Journal 1999; 77: 161–163.
- Lamb CR, Grierson J. Ultrasonographic appearance of primary gastric neoplasia in 21 dogs. Journal of Small Animal Practice 1999; 40: 211–215.
- Sautter JH, Hanlon GF. Gastric neoplasms in the dog: a report of 20 cases. Journal of the American Veterinary Medical Association 1975; 166: 691–696.
- Gallay J, Belanger MC, Helie P et al. Cardiac leiomyoma associated with advanced atrioventricular block in a young dog. Journal of Veterinary Cardiology 2011; 13: 71–77.
- Blackwood L, Sullivan M, Thompson H. Urethral leiomyoma causing post renal failure in a bitch. The Veterinary Record 1992; 131: 416–417.
- Heng HG, Lowry JE, Boston S et al. Smooth muscle neoplasia of the urinary bladder wall in three dogs. Veterinary Radiology & Ultrasound 2006; 47: 83–86.
- Laluha P, Grest P, Eichenberger S et al. Leiomyoma of a kidney in a dog: a rare diagnosis. Schweizer Archiv fur Tierheilkunde 2006; 148: 303–307.
- Mills JH, Moore JT, Orr JP. Canine renal leiomyoma–an unusual tumour. Canadian Veterinary Journal 1977; 18: 76–78.
- Sontas BH, Ozyogurtcu H, Turna O et al. Uterine leiomyoma in a spayed poodle bitch: a case report. Reproduction in Domestic Animals 2010; 45: 550–554.
- Eker K, Salmanoglu MR, Vural SA. Unilateral leiomyoma in the mesosalpinx of a dog. Journal of the American Animal Hospital Association 2006; 42: 392–394.
- Weissman A, Jimenez D, Torres B et al. Canine vaginal leiomyoma diagnosed by CT vaginourethrography. Journal of the American Animal Hospital Association 2013; 49: 394–397.
- Patnaik AK, Liu SK. Leiomyoma of the tunica vaginalis in a dog. Cornell Veterinarian 1975; 65: 228–231.
- Black AP, Liu S, Randolph JF. Primary tracheal leiomyoma in a dog. Journal of the American Veterinary Medical Association 1981; 179: 905–907.
- Carpenter JL, Hamilton TA. Angioleiomyoma of the nasopharynx in a dog. Veterinary Pathology 1995; 32: 721–723.
- Boari A, Barreca A, Bestetti GE et al. Hypoglycemia in a dog with a leiomyoma of the gastric wall producing an insulin-like growth factor II-like peptide. European Journal of Endocrinology 1995; 132: 744–750.
- Beaudry D, Knapp DW, Montgomery T et al. Hypoglycemia in four dogs with smooth muscle tumors. Journal of Veterinary Internal Medicine 1995; 9: 415–418.
- Bagley RS, Levy JK, Malarkey DE. Hypoglycemia associated with intra-abdominal leiomyoma and leiomyosarcoma in six dogs. Journal of the American Veterinary Medical Association 1996; 208: 69–71.
- Billson FM, Mould JR, Lee WR. Myxoid leiomyoma of the iris in a dog. Veterinary Ophthalmology 2003; 6: 173–176.
- Arat YO, Font RL, Chaudhry IA et al. Leiomyoma of the orbit and periocular region: a clinicopathologic study of four cases. Ophthalmic Plastic and Reconstructive Surgery 2005; 21: 16–22.
- Lee V, Azari AA, Nehls S et al. Leiomyoma of the lower eyelid. JAMA Ophthalmology 2013; 131: 1085.
- Hui JI, Buchser NM, Dubovy SR. Primary eyelid leiomyoma. Ophthalmic Plastic and Reconstructive Surgery 2011; 27: e102–e103.
- Brannan SO, Cheung D, Trotter S et al. A conjunctival leiomyoma. American Journal of Ophthalmology 2003; 136: 749– 750.
- Tuncer S, Peksayar G, Demiryont M et al. Longterm follow-up of a patient with iris leiomyoma treated with partial lamellar iridocyclectomy. Acta Ophthalmologica Scandinavica 2004; 82: 112– 114.
- Cornand G, Brisou B, Cozette P et al. A case of leiomyoma of the iris. Archives D’ophtalmologie et Revue Generale D’ophtalmologie 1975; 35: 245–252.
- Oh KJ, Kwon BJ, Han MH et al. MR imaging findings of uveal leiomyoma: three cases. American Journal of Neuroradiology 2005; 26: 100–103.
- Kanavi MR, Soheilian M, Peyman GA. Ciliary body leiomyoma with atypical features. Canadian Journal of Ophthalmology 2007; 42: 336–337.
- Kiratli H, Bilgic S, Soylemezoglu F. Ciliary body leiomyomas. Three case reports. Journal Francais D’Ophtalmologie 2005; 28: 1105–1109.
- Odashiro AN, Fernandes BF, Al-Kandari A et al. Report of two cases of ciliary body mesectodermal leiomyoma: unique expression of neural markers. Ophthalmology 2007; 114: 157–161.
- Shields JA, Shields CL, Eagle RC Jr. Mesectodermal leiomyoma of the ciliary body managed by partial lamellar iridocyclochoroidectomy. Ophthalmology 1989; 96: 1369–1376.
- Miyamoto K, Kashii S, Oishi A et al. Mesectodermal leiomyoma confined to the posterior choroid. Japanese Journal of Ophthalmology 2007; 51: 240–243.
- Perri P, Paduano B, Incorvaia C et al. Mesectodermal leiomyoma exclusively involving the posterior choroid. American Journal of Ophthalmology 2002; 134: 451–454.
- Shields JA, Shields CL, Eagle RC Jr et al. Observations on seven cases of intraocular leiomyoma The 1993 Byron Demorest Lecture Archives of Ophthalmology 1994; 112: 521–528
- Shields JA, Shields CL Observations on intraocular leiomyomas Transactions – Pennsylvania Academy of Ophthalmology and Otolaryngology 1990; 42: 945–950
- Foss AJ, Pecorella I, Alexander RA et al. Are most intraocular “leiomyomas” really melanocytic lesions? Ophthalmology 1994; 101: 919–924
- Lai CT, Tai MC, Liang CM et al. Unusual uveal tract tumor: mesectodermal leiomyoma of the ciliary body Pathology International 2004; 54: 337–342
- Maas CP, ter Haar G, van der Gaag I et al. Reclassification of small intestinal and cecal smooth muscle tumors in 72 dogs: clinical, histologic, and immunohistochemical evaluation Veterinary Surgery 2007; 36: 302–313
- Saito A, Kotani T Estimation of lacrimal level and testing methods on normal beagles Veterinary Ophthalmology 2001; 4: 7–11
- Slatter DH Textbook of Small Animal Surgery, Vol 2, Saunders Elsevier, Philadelphia, 2003; Chapter 90, 1366
- Sato T, Yamamoto A, Shibuya H et al. Intraocular peripheral nerve sheath tumor in a dog Veterinary Ophthalmology 2005; 8: 283–286
- Pumarola M, Anor S, Borras D et al. Malignant epithelioid schwannoma affecting the trigeminal nerve of a dog Veterinary Pathology 1996; 33: 434–436
- Sawamoto O, Yamate J, Kuwamura M et al. A canine peripheral nerve sheath tumor including peripheral nerve fibers Journal of Veterinary Medical Science 1999; 61: 1335–1338
- Kircher CH, Garner FM, Robinson FR Tumours of the eye and adnexa Bulletin of the World Health Organization 1974; 50: 135–142
- Bettini G, Morini M, Marcato PS Gastrointestinal spindle cell tumours of the dog: histological and immunohistochemical study Journal of Comparative Pathology 2003; 129: 283–293
- Saito A, Izumisawa Y, Yamashita K et al. The effect of third eyelid gland removal on the ocular surface of dogs Veterinary Ophthalmology 2001; 4: 13–18
© 2015 American College of Veterinary Ophthalmologists, Veterinary Ophthalmology, 19, 347–354
